
Data Integrity
What is data integrity?
Data integrity means that your data is complete, consistent, and trustworthy throughout its lifecycle — from generation to archival per ALCOA+ Principles. These principles are not optional — they are embedded expectations in global GMP regulations.In cGMP environments, data is not just information—it is documented evidence that processes were executed as intended and that products meet predefined quality standards. Regulators such as the U.S. Food and Drug Administration and Pharmaceutical Inspection Co-operation Scheme expect organisations to demonstrate that all data is trustworthy, traceable, and protected from unauthorised alteration.
Why is it vital in GMP?
Data integrity is critical because every GMP decision is based on data. Batch release, stability conclusions, process validation, and clinical outcomes all depend on the accuracy and completeness of recorded information. If the underlying data is unreliable, then the decisions derived from it cannot be justified. Regulators therefore consider data integrity failures as indicators of broader weaknesses in the quality system, often linking them to potential risks to patient safety and product quality.
How do you maintain data integrity?
Maintaining data integrity requires a holistic and lifecycle-based approach that integrates technology, procedures, and organisational culture. This includes implementing validated computerised systems with secure access controls, ensuring audit trails are enabled and regularly reviewed, and establishing clear procedures for data generation, processing, and archival. Equally important is fostering a quality culture where personnel are trained to record data accurately and contemporaneously, and where transparency is encouraged. Data integrity is sustained not only through controls, but through consistent oversight and accountability across the organisation.
Can data be tampered with?
Yes, data can be intentionally or unintentionally altered if appropriate controls are not in place. Inadequate system configurations, excessive user privileges, lack of audit trails, or reliance on uncontrolled tools such as spreadsheets can create opportunities for data manipulation. Regulators assume that data can be tampered with unless systems are designed to prevent, detect, and record such actions. This is why robust technical controls, procedural safeguards, and independent review mechanisms are essential in regulated environments.
Who is responsible for data integrity?
Data integrity is a shared responsibility across the organisation, but accountability is clearly defined. Operators and analysts are responsible for accurate and timely data entry, supervisors ensure proper review and oversight, and the Quality Unit is responsible for governance, compliance, and system robustness. Senior management holds ultimate responsibility for establishing a culture of integrity and ensuring that adequate resources, systems, and controls are in place. Regulators increasingly assess not just individual compliance, but organisational accountability and leadership commitment.
What happens if data integrity is compromised?
It risks product recalls, regulatory penalties, and potential harm to consumers. When data integrity is compromised, the impact extends far beyond a single record or event. The reliability of affected data is questioned, which may invalidate batch release decisions, analytical results, or entire studies. Regulatory authorities may escalate inspections, expand the scope of investigations, and impose enforcement actions such as warning letters, import alerts, or batch holds. In addition to regulatory consequences, organisations face significant business risks, including delayed approvals, financial losses, and reputational damage. Ultimately, compromised data integrity undermines trust—both with regulators and with patients.
In cGMP environments, data integrity is not simply about compliance—it is about credibility. If your data cannot be trusted, regulators will conclude that your processes—and your product—cannot be trusted either.
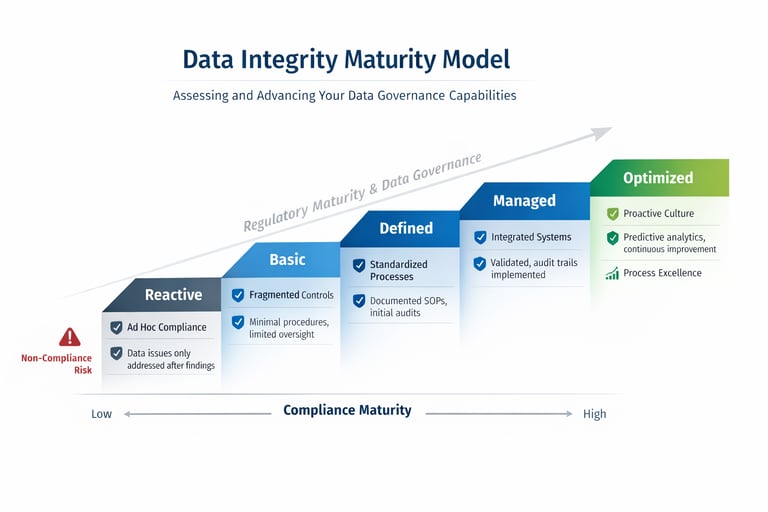
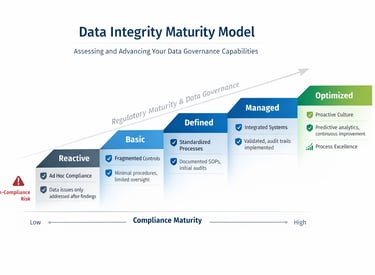
Why Data Integrity Matters
Core Principles of Data Integrity (ALCOA+)
The foundation of trustworthy, inspection-ready data
Regulators globally, including the U.S. Food and Drug Administration, European Medicines Agency, and Pharmaceutical Inspection Co-operation Scheme, expect organisations to demonstrate that all GMP data adheres to the ALCOA+ principles.
These principles define whether your data can be trusted during regulatory review.
Because Data is Your Product’s Story — and Regulators Read Every Line
In cGMP-regulated environments, data integrity is not just a compliance requirement — it is the foundation of product quality, patient safety, and regulatory trust. Every decision made during development, manufacturing, testing, and release is only as reliable as the data that supports it.
Regulatory agencies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and Pharmaceutical Inspection Co-operation Scheme (PIC/S) have repeatedly emphasised that data integrity failures are among the most critical and frequently cited GMP violations globally.
Attributable
Every data point must clearly identify who performed the action and when.
👉 No anonymous entries, shared logins, or undocumented changes.
Legible
Data must be readable, permanent, and understandable throughout its lifecycle.
👉 Applies to both handwritten and electronic records.
Contemporaneous
Data must be recorded at the time the activity is performed.
👉 Backdating or delayed recording is a critical violation.
Original
The first capture of data (or a certified true copy) must be retained.
👉 Raw data must always be available for review.
Accurate
Data must be correct, complete, and free from errors.
👉 Any corrections must be justified, traceable, and documented.
Complete
All data — including failed runs, deviations, and repeats — must be retained.
👉 Selective reporting is a major regulatory concern.
Consistent
Data must follow a logical and chronological sequence.
👉 Time stamps, sequence of events, and audit trails must align.
Enduring
Data must be recorded in a durable and permanent format.
👉 Temporary notes or unofficial records are not acceptable.
Available
Data must be readily accessible for review and inspection.
👉 Especially critical during audits and regulatory inspections.
Real-World Impact
Data integrity is not theoretical — it has direct regulatory and business consequences.
Common Regulatory Findings
Manipulated or deleted laboratory data
Disabled audit trails
Shared user credentials
Backdated or fabricated entries
Mismatch between raw and reported results
Regulatory Actions Observed
FDA Warning Letters
Import Alerts (products blocked from markets)
Clinical Holds
Product Recalls
License Delays or Rejections
👉 Explore actual enforcement cases: FDA Warning Letters Database
Note: Open the link and type Data Integrity in the search box
Many warning letters explicitly cite systemic data manipulation and governance failures — not just isolated errors.
Your Quality & CGMP Consulting Expert
20 Cecil Street, #14-00, PLUS, Singapore 049705
Email: contact@biocellgene.com
© 2026 BioCellGene. A division of Mindcube Consulting Pte. Ltd. All rights reserved.
BioCellGene










