GMP FACILITY DESIGN & READINESS
Overview
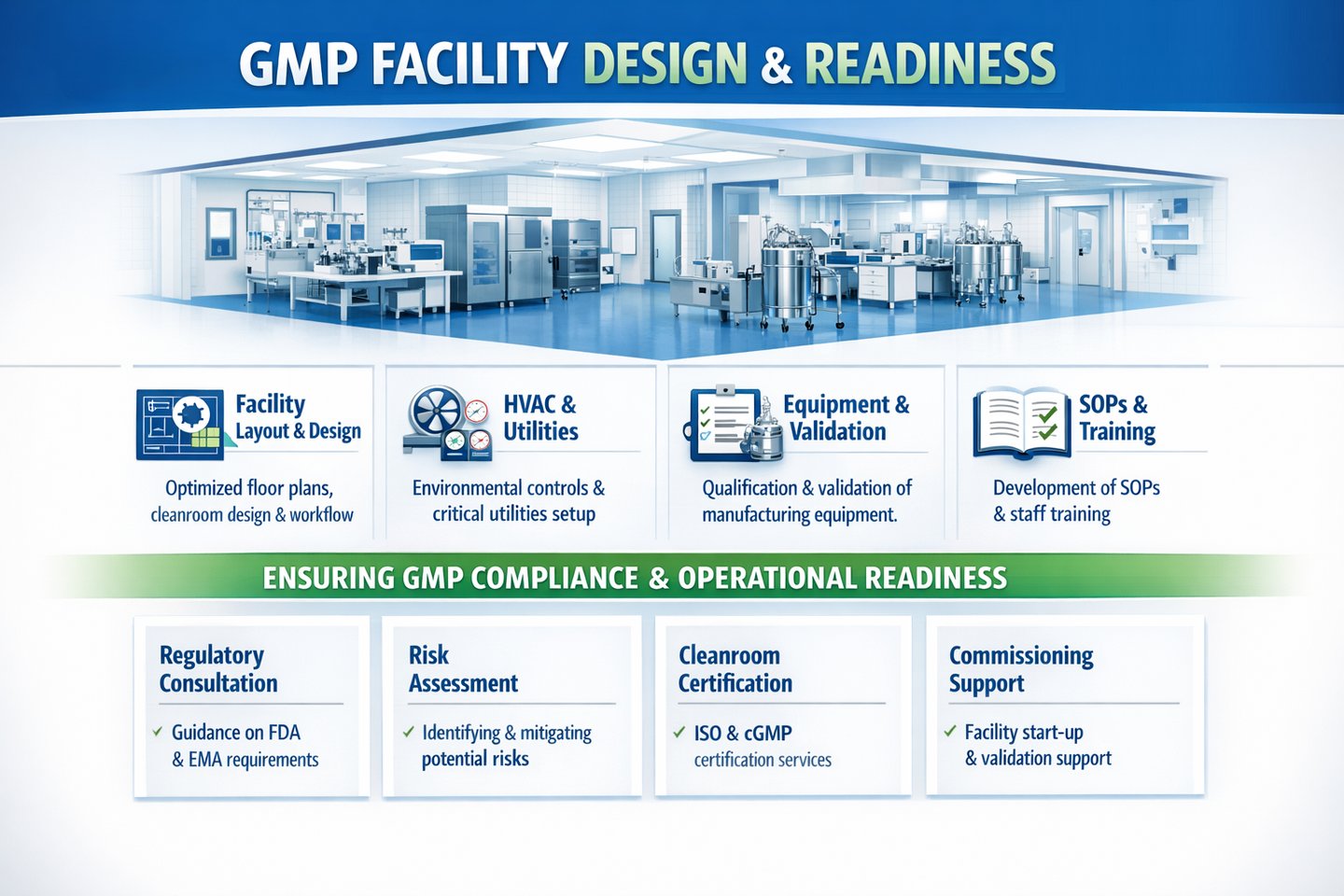
We support GMP facility design and readiness aligned with EU Annex 1 and global GMP expectations, ensuring optimal flow, contamination control, and operational efficiency.
Core Deliverables
Facility layout and zoning strategy
Personnel and material flow design
Cleanroom classification and pressure cascade
Contamination Control Strategy (CCS)
Environmental monitoring program design
Utilities qualification strategy
Regulatory Framework
EU GMP Annex 1 (2022)
PIC/S PE009
ISO 14644
Advanced Capabilities
Greenfield facility planning
Facility remediation for inspections
ATMP-specific facility design
Outcome
A compliant, efficient facility designed to minimise risk and support scalable operations.

Your Quality & CGMP Consulting Expert
20 Cecil Street, #14-00, PLUS, Singapore 049705
Email: contact@biocellgene.com
© 2026 BioCellGene. A division of Mindcube Consulting Pte. Ltd. All rights reserved.
BioCellGene

